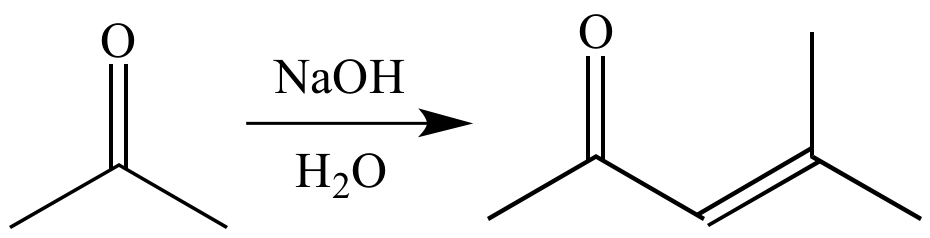
Predict what compound(s) might result from the base-catalyzed condensation of acetone with acetaldehyde. | Homework.Study.com

List all possible products if acetaldehyde (ethanal) was reacted with acetone in an aldol condensation. | Homework.Study.com

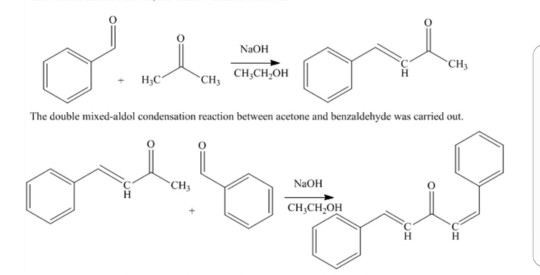
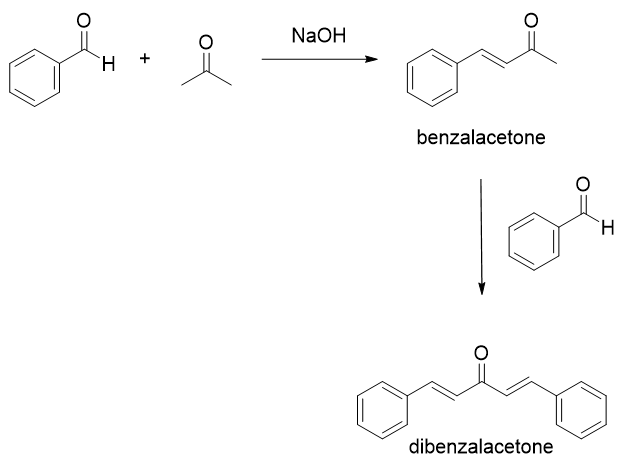
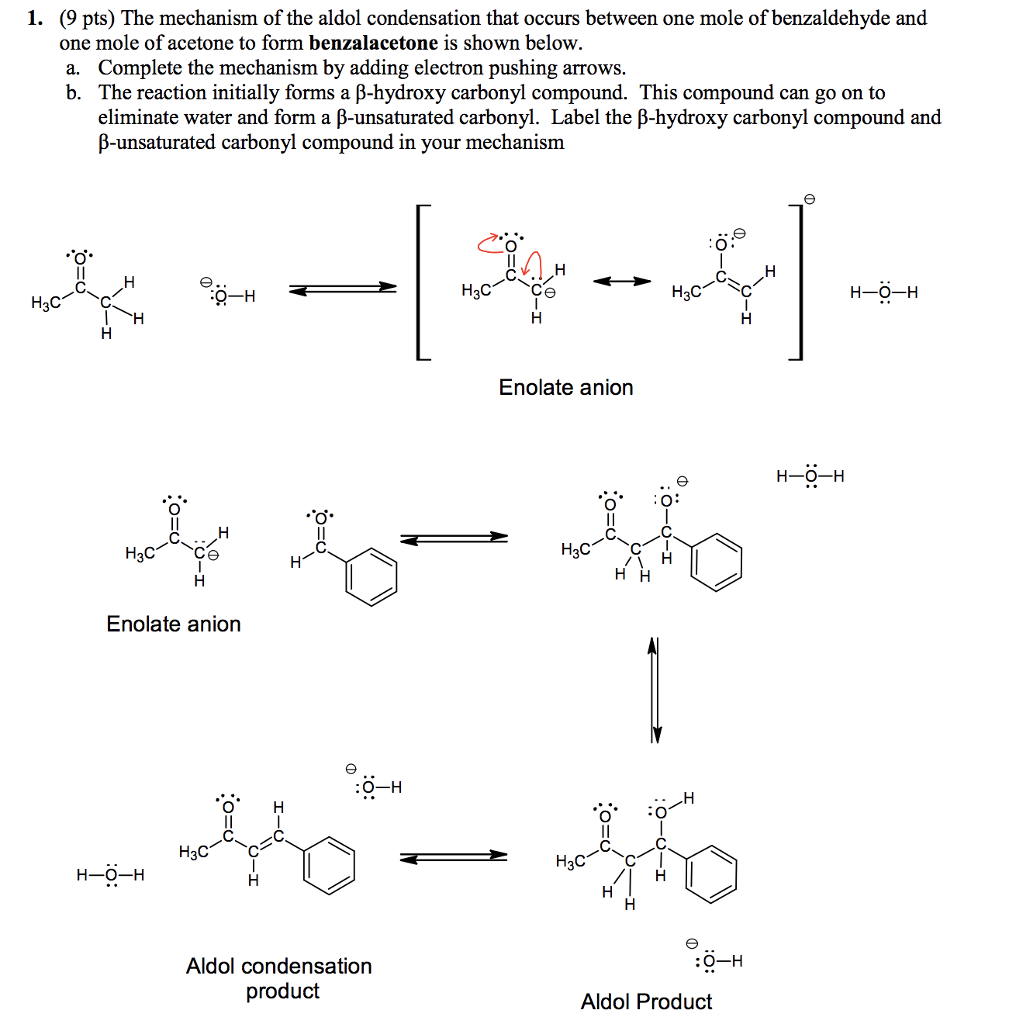
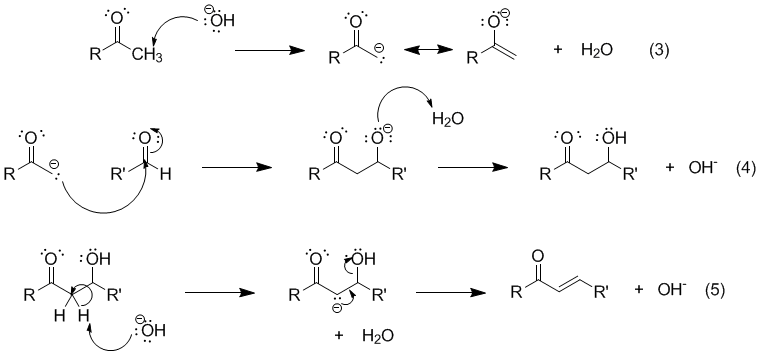
Write out all the steps in the mechanism for the base catalyzed condensation of acetone with 2 mol of benzaldehyde. | Homework.Study.com

Why Benzaldehyde and acetone can't do self-condensation in claisen-Schmidt reaction condensation? 2)In the second step of the same reaction, why do we add ethanol to form dibenzalideneacetone? | Homework.Study.com
![PDF] Reaction Efficiency of Crossed-Aldol Condensation between Acetone and Benzaldehyde over ZrO2 and ZrO2-Montmorillonite Catalyst | Semantic Scholar PDF] Reaction Efficiency of Crossed-Aldol Condensation between Acetone and Benzaldehyde over ZrO2 and ZrO2-Montmorillonite Catalyst | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0a54e47c87151b8af017126cb18d23eebb7b47e5/4-Figure4-1.png)
PDF] Reaction Efficiency of Crossed-Aldol Condensation between Acetone and Benzaldehyde over ZrO2 and ZrO2-Montmorillonite Catalyst | Semantic Scholar
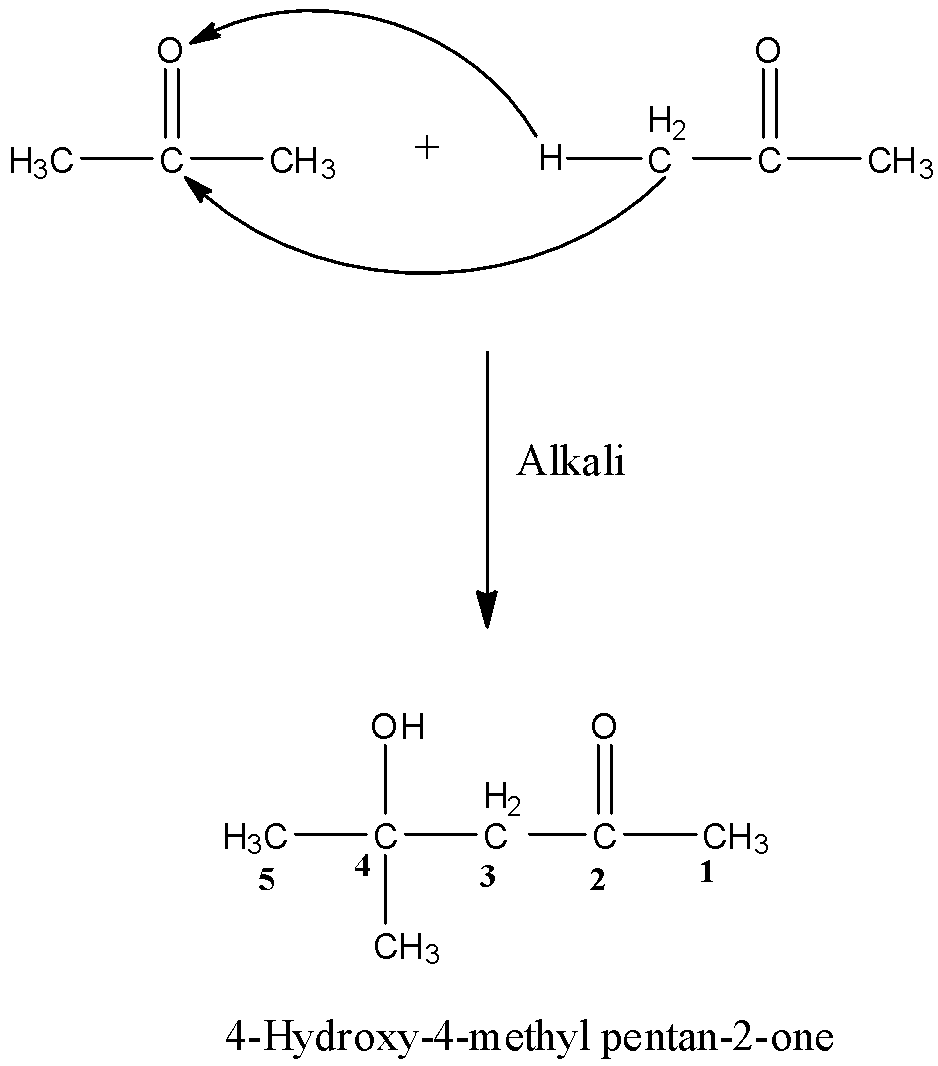
Aldol condensation product of acetone on dehydration gives:(a)- But-2-enal(b)- 2–Methyl-pent–3–en–4–one(c)- 4–Hydroxy–4–methyl pentan–2 –one (d)- 4–Methyl–pent–3–en–2–one

Acetone can undergo aldol condensation with itself. Select the correct product from structures A-E. | Homework.Study.com
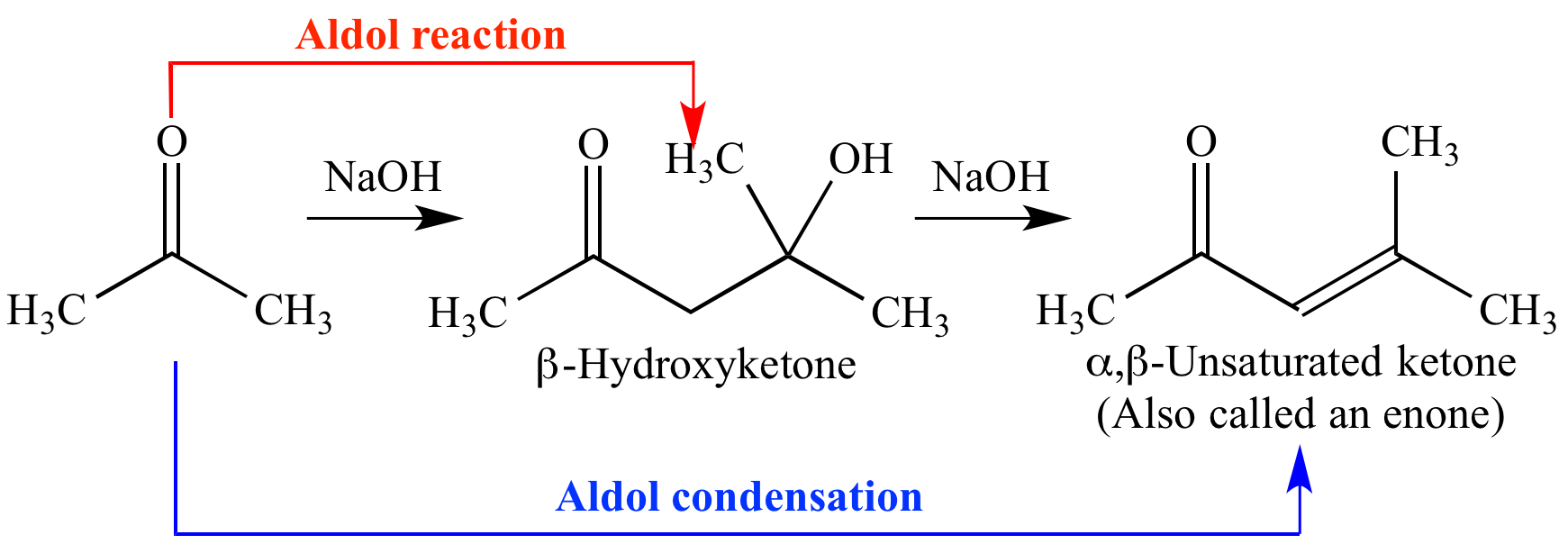
Illustrated Glossary of Organic Chemistry - Aldol Condensation; Crossed Aldol Condensation; Claisen-Schmidt Condensation

Self-Condensation of Acetone of Strontium Stannate under Supercritical Conditions | Russian Journal of Physical Chemistry B

Explain aldol condensation reaction with acetone as the substrate - Chemistry - Aldehydes Ketones and Carboxylic Acids - 15276041 | Meritnation.com

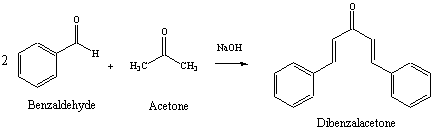
Benzylidene acetone is the product of mixed condensation between benzaldehyde and acetone. What is its structure?
![Benzalacetone is the product of mixed aldol condensation between benzaldehyde $({C_6}{H_5}CH = O)$ and acetone $[{(C{H_3})_2}C\\,\\, + \\,\\,O]$ . What is its structure?A) ${C_6}{H_5}CH = CHC(O)(C{H_3})$ B) ${C_6}{H_5}CH = C{(C{H_3})_2}$ C) ${C_6}{H_5 ... Benzalacetone is the product of mixed aldol condensation between benzaldehyde $({C_6}{H_5}CH = O)$ and acetone $[{(C{H_3})_2}C\\,\\, + \\,\\,O]$ . What is its structure?A) ${C_6}{H_5}CH = CHC(O)(C{H_3})$ B) ${C_6}{H_5}CH = C{(C{H_3})_2}$ C) ${C_6}{H_5 ...](https://www.vedantu.com/question-sets/c14e38d4-5c9a-41d9-83ce-33f5e5ac3f366187549734456439828.png)