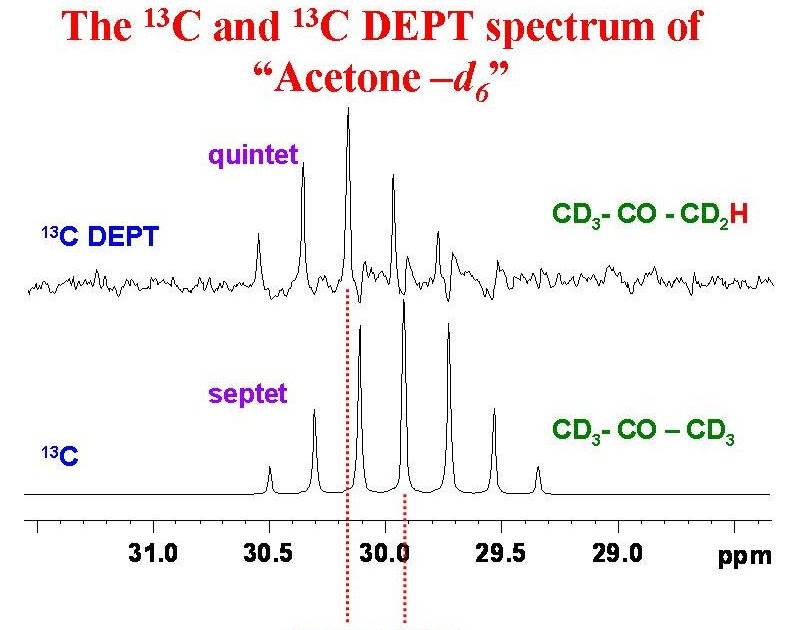
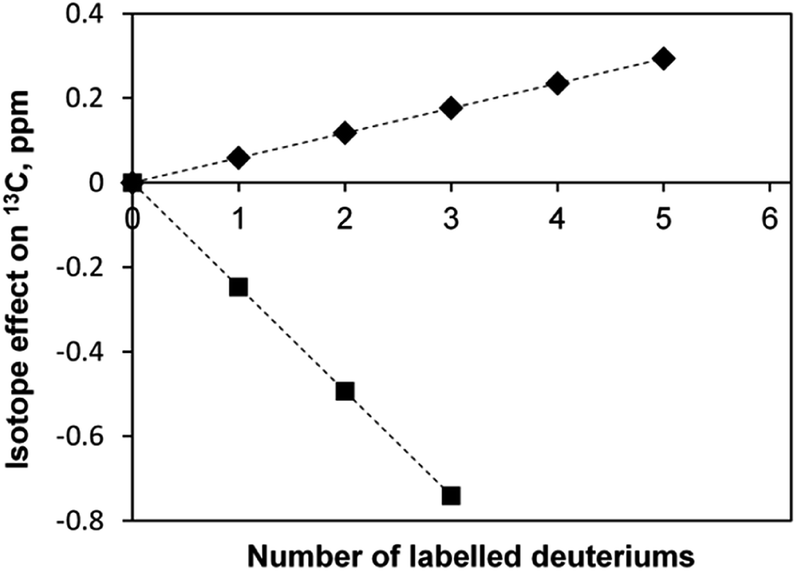
Molecules | Free Full-Text | On the Use of Deuterated Organic Solvents without TMS to Report 1H/13C NMR Spectral Data of Organic Compounds: Current State of the Method, Its Pitfalls and Benefits,

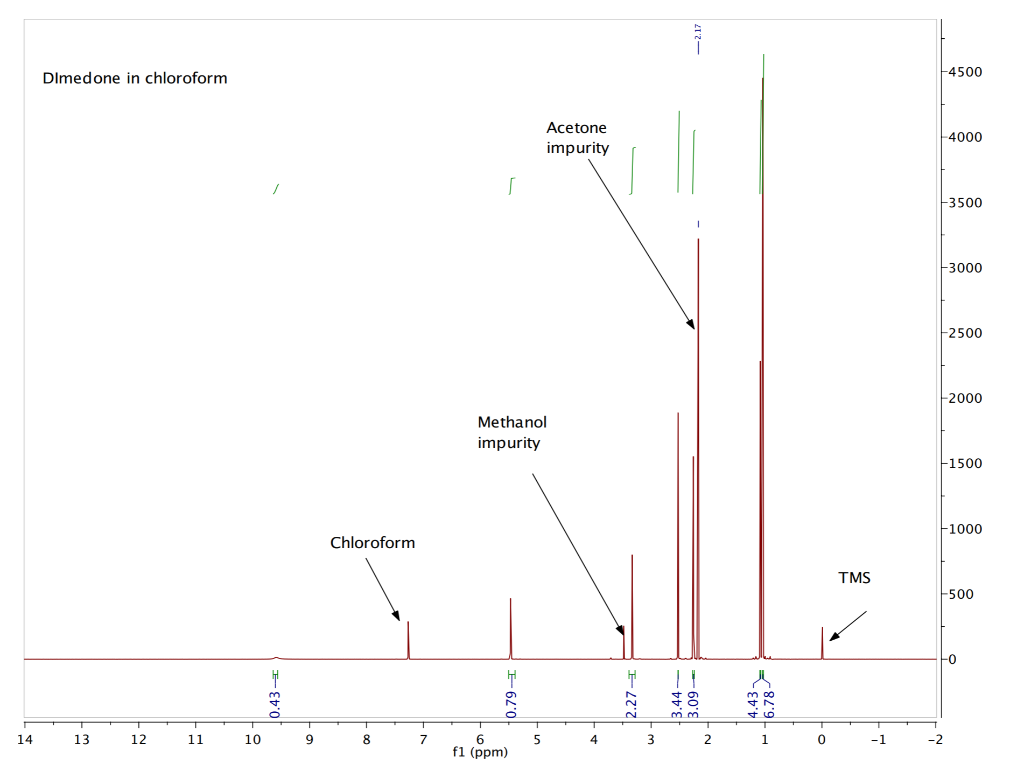
NMR Chemical Shifts of Trace Impurities: Industrially Preferred Solvents Used in Process and Green Chemistry | Organic Process Research & Development
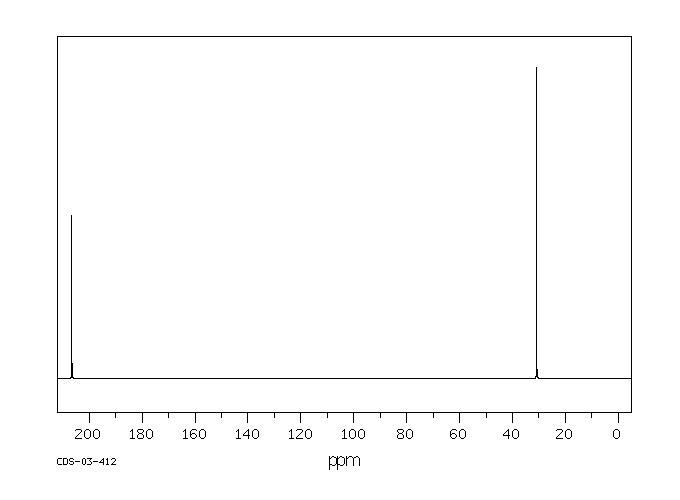
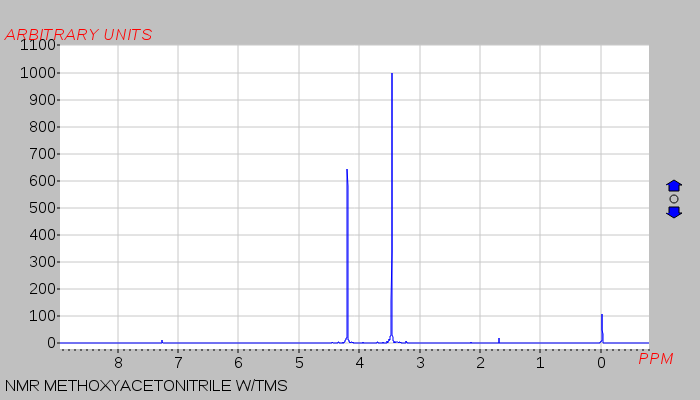
Proton–deuterium exchange of acetone catalyzed in imidazolium-based ionic liquid–D 2 O mixtures - RSC Advances (RSC Publishing) DOI:10.1039/D0RA04206D

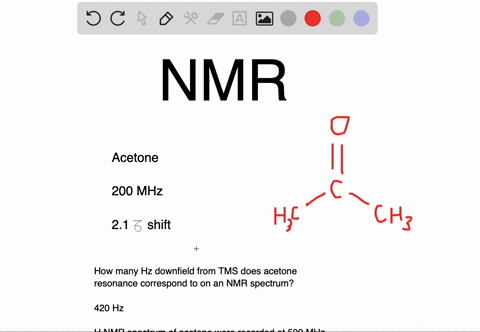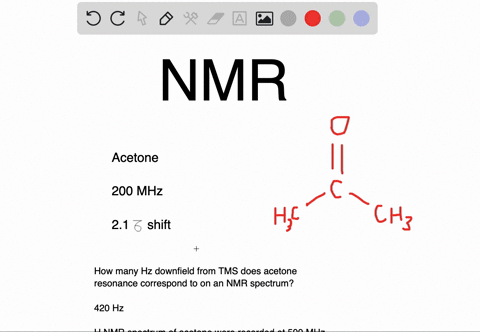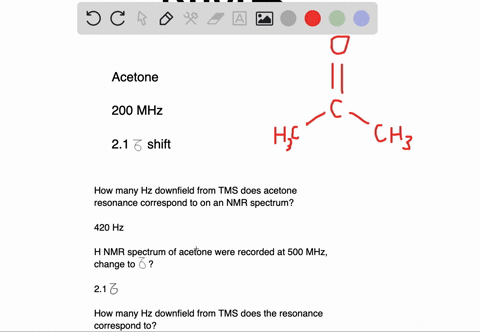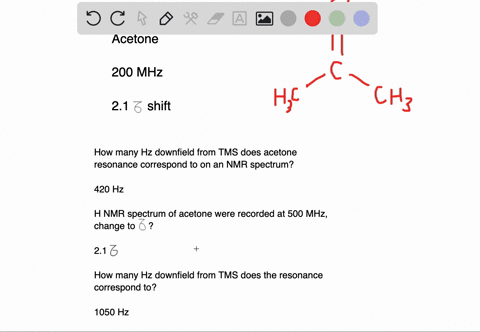
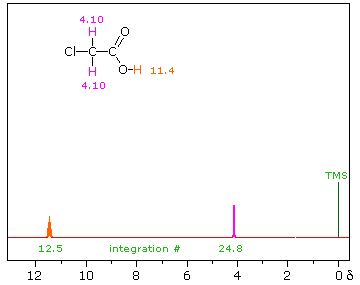
Draw the NMR spectrum for each of the following compounds. Draw the peaks in the approximate positions where they would appear in a recorded spectrum. Also indicate the splitting pattern of the

NMR Chemical Shifts of Trace Impurities: Industrially Preferred Solvents Used in Process and Green Chemistry - ScienceDirect

Draw the NMR spectrum for each of the following compounds. Draw the peaks in the approximate positions where they would appear in a recorded spectrum. Also indicate the splitting pattern of the

NMR Chemical Shifts of Trace Impurities: Industrially Preferred Solvents Used in Process and Green Chemistry | Organic Process Research & Development
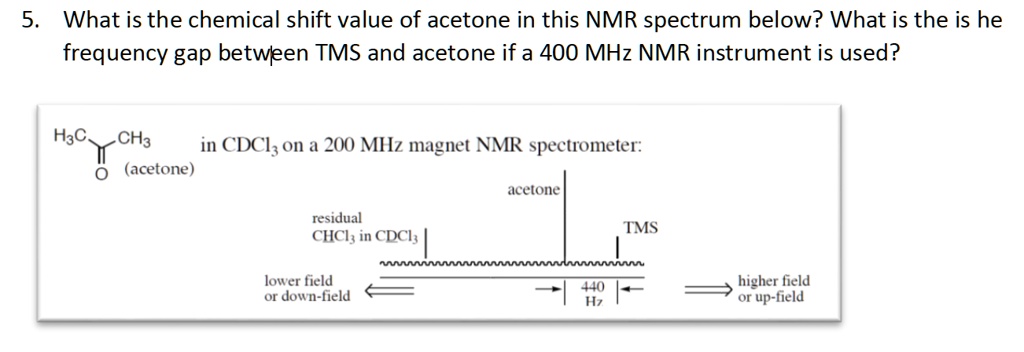
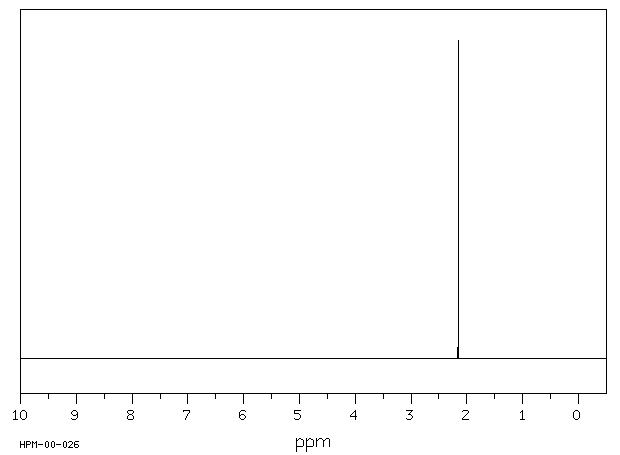
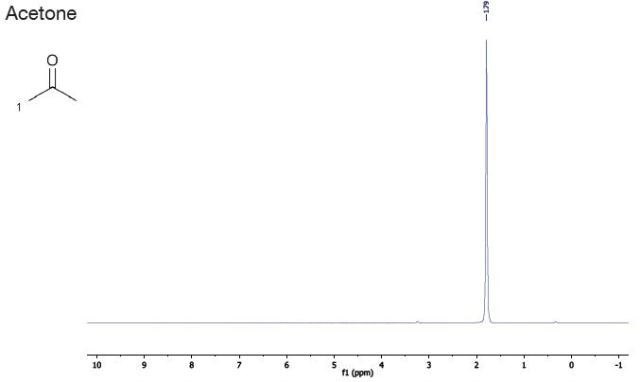
SOLVED: What is the chemical shift value of acetone in this NMR spectrum below? What is the frequency gap between TMS and acetone if a 400 MHz NMR instrument is used? H3C

![Chemical shifts of the ring protons of lactose [15 mM] in acetone d6... | Download Table Chemical shifts of the ring protons of lactose [15 mM] in acetone d6... | Download Table](https://www.researchgate.net/publication/10842958/figure/tbl3/AS:601744492990471@1520478475394/Chemical-shifts-of-the-ring-protons-of-lactose-15-mM-in-acetone-d6-water-mixture-at.png)