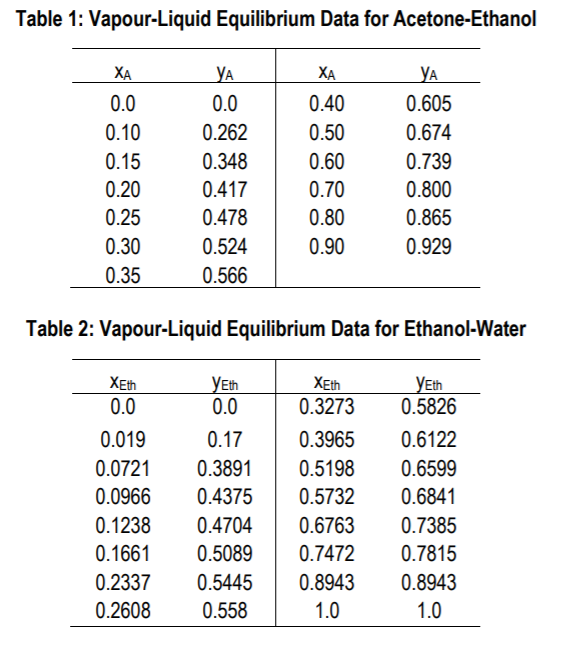
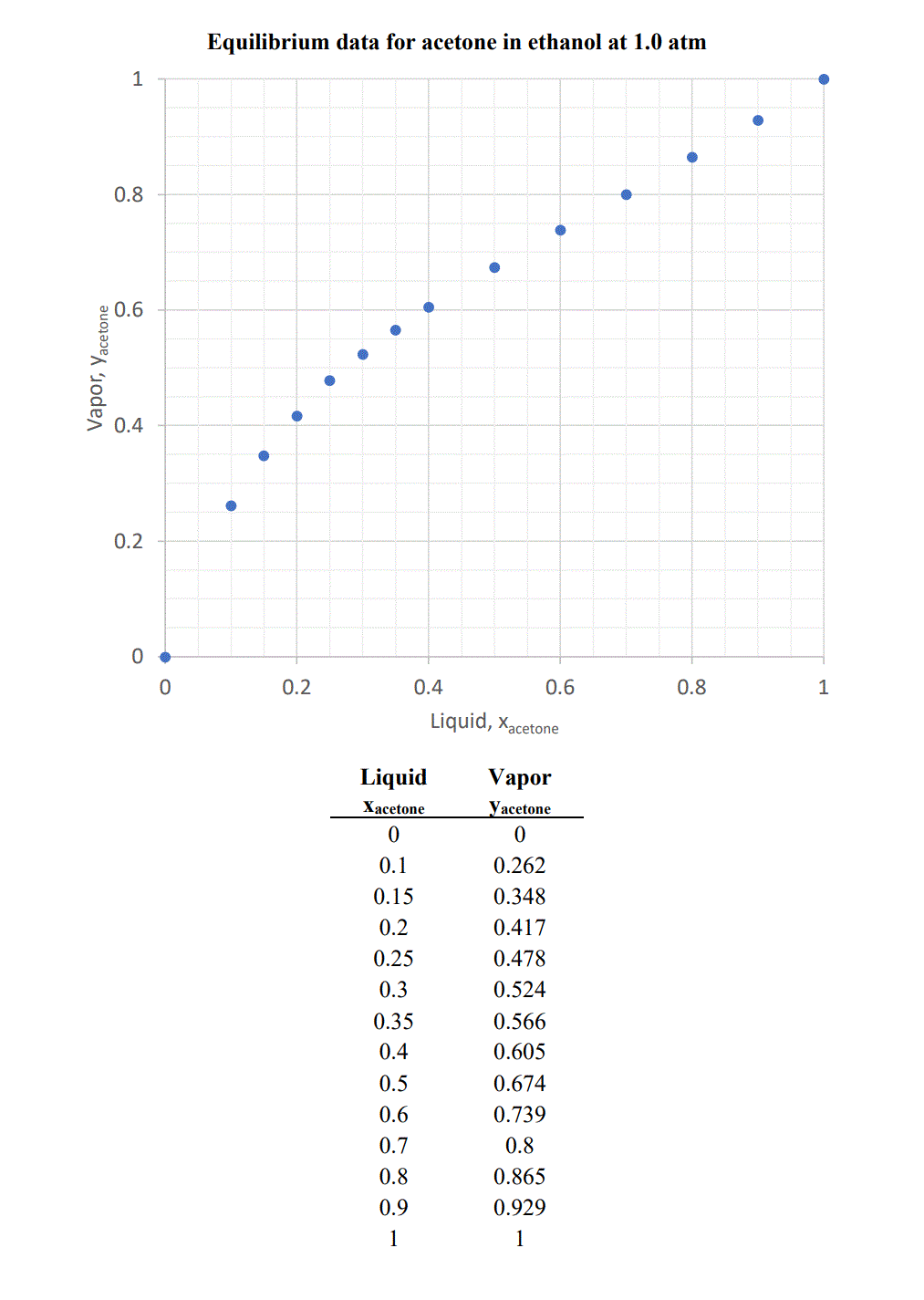
Isobaric vapor-liquid equilibrium for acetone + methanol system containing different ionic liquids at 101.3 kPa
![PDF] Liquid-Liquid Equilibrium Data for Butan-2-ol - Ethanol - Water, Pentan-1-ol - Ethanol - Water and Toluene - Acetone - Water Systems | Semantic Scholar PDF] Liquid-Liquid Equilibrium Data for Butan-2-ol - Ethanol - Water, Pentan-1-ol - Ethanol - Water and Toluene - Acetone - Water Systems | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c1fa0b8a36ef8526c29d6a283bd62f1c9d37340c/2-TableI-1.png)
PDF] Liquid-Liquid Equilibrium Data for Butan-2-ol - Ethanol - Water, Pentan-1-ol - Ethanol - Water and Toluene - Acetone - Water Systems | Semantic Scholar

Vapor–Liquid Equilibrium Data for Binary Mixtures of Acetic Acid + Anisole, Acetone + Anisole, and Isopropanol + Anisole at Pressure 96.15 kPa | Journal of Chemical & Engineering Data

Effects of Diphenylamine on Binary Systems of Acetone + Ethanol, Acetone + Cyclohexane, and Ethanol + Cyclohexane at 101.3 kPa: Vapor–Liquid Equilibrium Measurement and Molecular Simulation | Journal of Chemical & Engineering Data
Vapor-liquid equilibria for binary and ternary systems composed of acetone, 1-butanol, and ethanol at atmospheric pressure

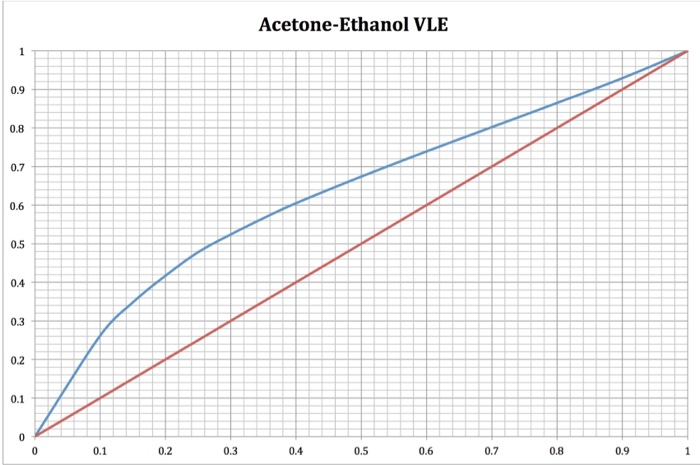
Plot of VLE data of the binary: acetone(1) + ethanol(2), iso-p ≈ 101.32... | Download Scientific Diagram

Isobaric vapor–liquid and vapor–liquid–liquid equilibrium data for the system water + ethanol + cyclohexane | Semantic Scholar

Vapor–Liquid Equilibrium Data for Binary Mixtures of Acetic Acid + Anisole, Acetone + Anisole, and Isopropanol + Anisole at Pressure 96.15 kPa | Journal of Chemical & Engineering Data
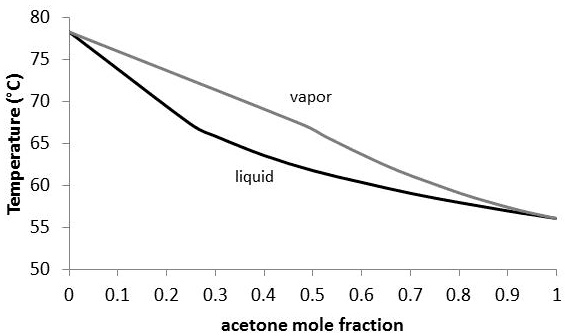
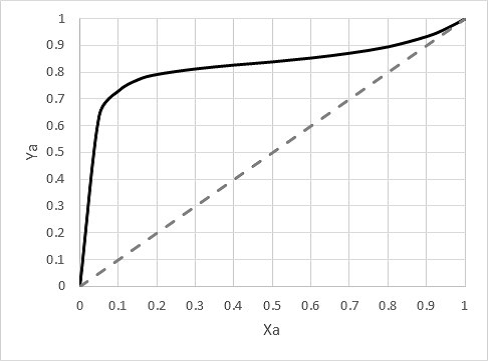
SOLVED: A vapor-liquid equilibrium diagram for acetone and ethanol at 1 atm is shown below. Estimate the mole fractions of acetone in the vapor and liquid phases at 65°C. The correct answer
![PDF] Liquid-Liquid Equilibrium Data for Butan-2-ol - Ethanol - Water, Pentan-1-ol - Ethanol - Water and Toluene - Acetone - Water Systems | Semantic Scholar PDF] Liquid-Liquid Equilibrium Data for Butan-2-ol - Ethanol - Water, Pentan-1-ol - Ethanol - Water and Toluene - Acetone - Water Systems | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c1fa0b8a36ef8526c29d6a283bd62f1c9d37340c/3-Figure3-1.png)