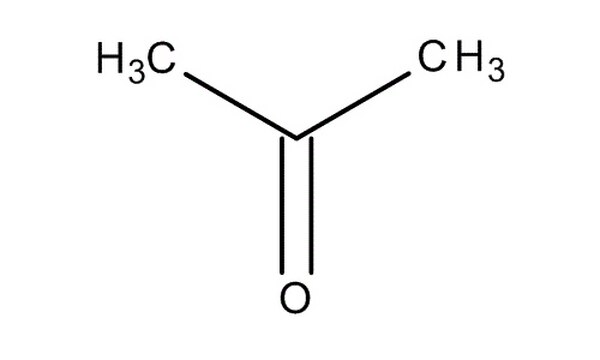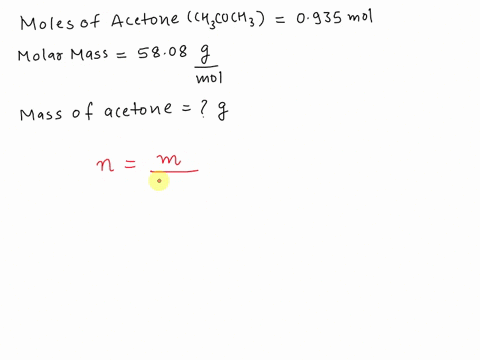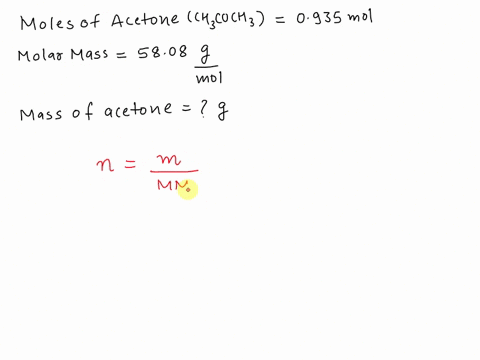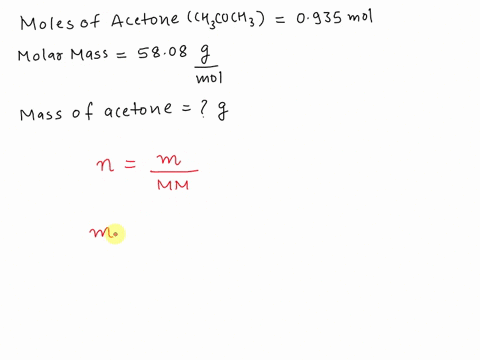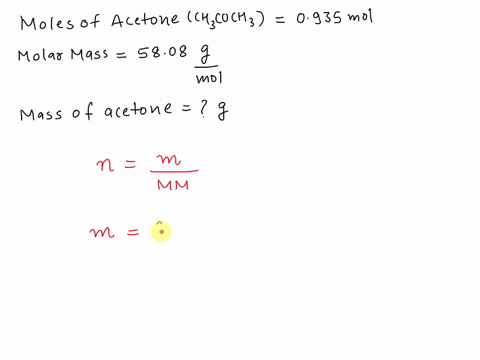
SOLVED: What is the mass in grams of 0.935 mol of acetone; CH;COCH3? The molar mass of acetone is 58.08 g/mol. A. 0.0161 g B. 54.3 g C. 57.1 g D. 62.1 g

SOLVED: A chemist wants to determine the molar mass of a naturally occurring substance isolated from spearmint. She adds 4.531 g of this substance to 25.00 g of acetone (C3H6O; molar mass

A student determines the molar mass of acetone, CH,COCH, by the method used in m found that the equilibrium temperature of a mixture of ice and water was 1.0°C on of a

SOLVED: What is the mass in grams of 0.935 mol of acetone; CH;COCH3? The molar mass of acetone is 58.08 g/mol. A. 0.0161 g B. 54.3 g C. 57.1 g D. 62.1 g

Acetone (Solvent ) Molar Mass at best price in Delhi by Satish Chemical (India) Private Limited | ID: 6758184888

SOLVED: What is the mass (in g) of 1.935 mol of acetone, CH3COCH3? The molar mass of acetone is 58.08 g*mol-1.

Acetone, or propanone, is an organic compound with the formula (CH3)2CO. It is the simplest and smallest ketone. It is … | Chemical structure, Chemistry, Molar mass
![Calculate molar mass of following compounds.i) [CO(NH2)2]ii) [CH3COCH3]iii) [H3BO3]iv) [H2SO4] - YouTube Calculate molar mass of following compounds.i) [CO(NH2)2]ii) [CH3COCH3]iii) [H3BO3]iv) [H2SO4] - YouTube](https://i.ytimg.com/vi/CCWl-z3Wdmo/hqdefault.jpg)
Calculate molar mass of following compounds.i) [CO(NH2)2]ii) [CH3COCH3]iii) [H3BO3]iv) [H2SO4] - YouTube

Define osmosis. A solution containing 1.21 g of camphor (molar mass 152 g mol-') in 26.68 g of acetone boils 329.95 K. The boiling point of pure acetone is 329.45 K. Calculate molal elevation constant acetone.
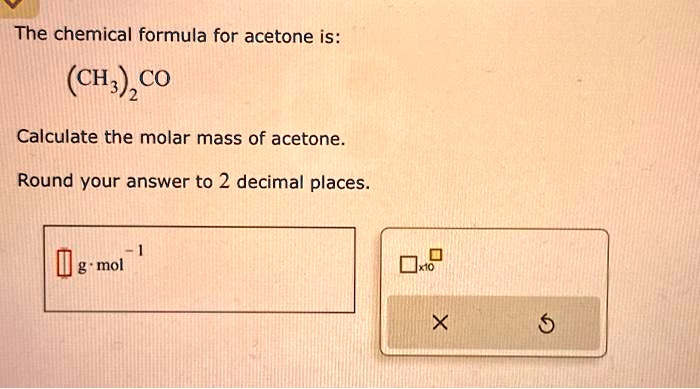
SOLVED: The chemical formula for acetone is: (CH),CO Calculate the molar mass of acetone Round your answer to 2 decimal places. g.mol









![Tamil] Calculate the molar mass of the following compounds. (ii) Ac Tamil] Calculate the molar mass of the following compounds. (ii) Ac](https://static.doubtnut.com/ss/web-overlay-thumb/3190387.webp)
![Acetone [(CH3)2CO] Molecular Weight Calculation - Laboratory Notes Acetone [(CH3)2CO] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/02/acetone-molecular-weight-calculation-300x200.jpg)