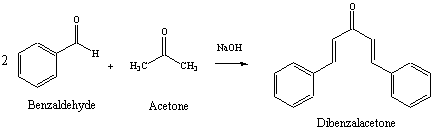
The reaction between benzaldehyde derivatives with acetone in presence... | Download Scientific Diagram

A reaction between benzaldehyde and propnaone and identification of the product. Explain fully the NMR, IR, CARBON SPECTRUM, HNMR, CNMR of the reaction of the mechanism. | Homework.Study.com
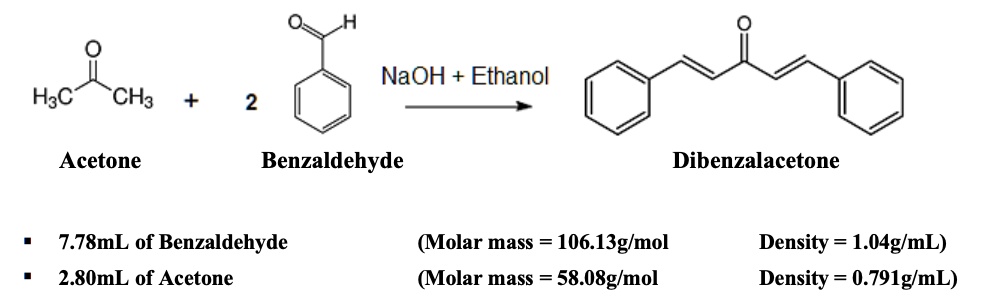
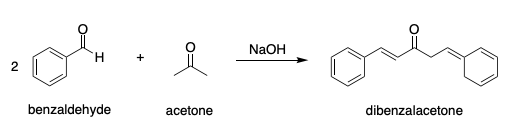
SOLVED: NaOH Ethanol HCl Acetone Benzaldehyde Dibenzalacetone 7.78 mL of Benzaldehyde 2.80 mL of Acetone (Molar mass = 106.13 g/mol) (Molar mass = 58.08 g/mol) Density = 1.04 g/mL Density = 0.791 g/mL CH3

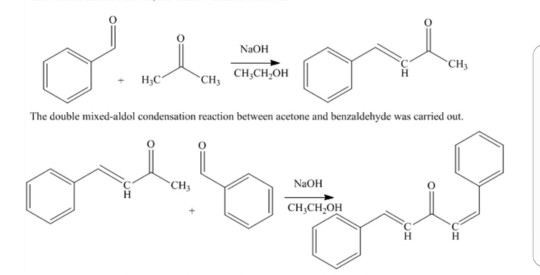
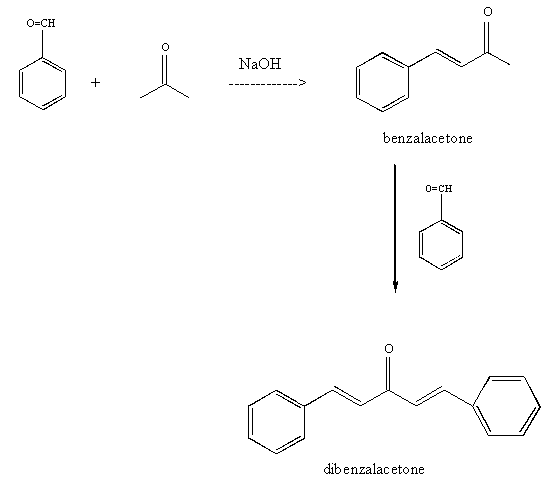
The product formed by aldol condensation between benzaldehyde and acetone is benzalacetone. Its structure is:
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone. - Sarthaks eConnect | Largest Online Education Community
![PDF] Reaction Efficiency of Crossed-Aldol Condensation between Acetone and Benzaldehyde over ZrO2 and ZrO2-Montmorillonite Catalyst | Semantic Scholar PDF] Reaction Efficiency of Crossed-Aldol Condensation between Acetone and Benzaldehyde over ZrO2 and ZrO2-Montmorillonite Catalyst | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0a54e47c87151b8af017126cb18d23eebb7b47e5/4-Figure4-1.png)
PDF] Reaction Efficiency of Crossed-Aldol Condensation between Acetone and Benzaldehyde over ZrO2 and ZrO2-Montmorillonite Catalyst | Semantic Scholar
Scheme 1 Mannich reaction between benzaldehyde 1, aniline 2 and acetone 3. | Download Scientific Diagram

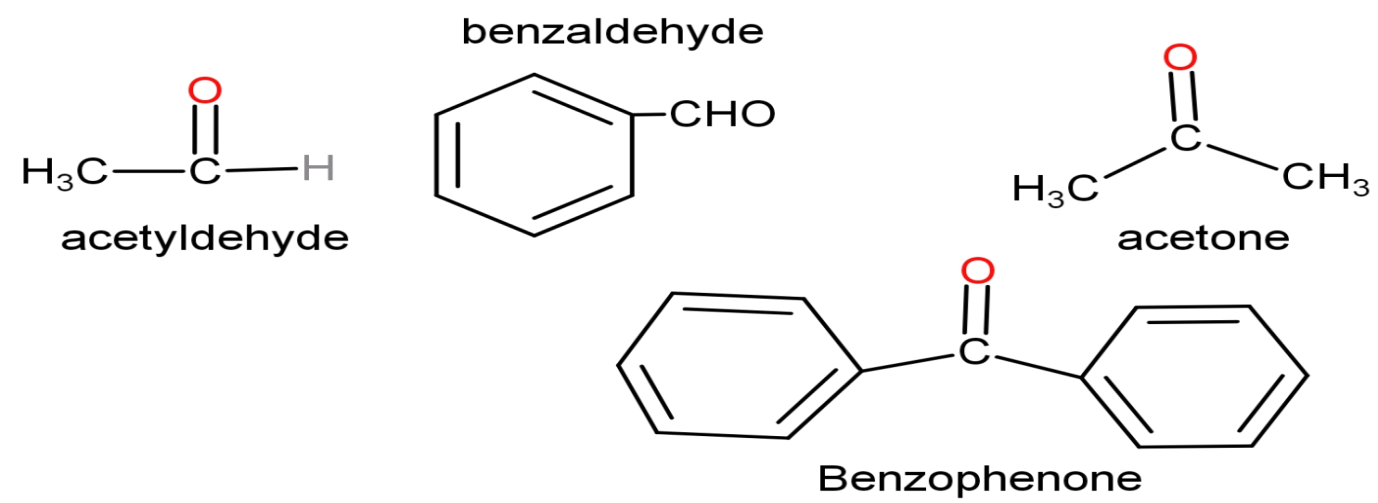
Highly tunable selectivity to benzaldehyde over Pd/ZrO2 catalysts in Oppenauer oxidation of benzyl alcohol using acetone as H-acceptor - ScienceDirect

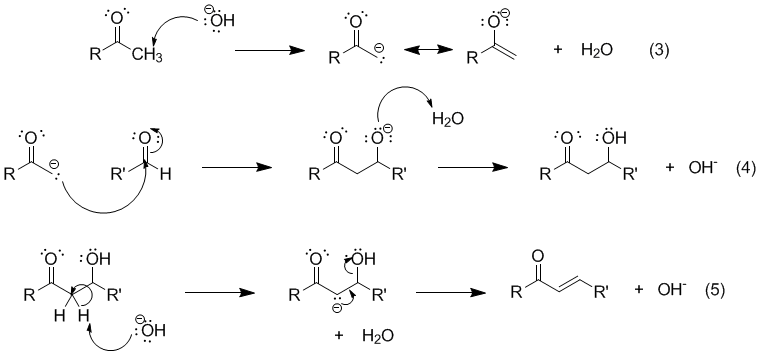
a) What is the step-wise mechanism for the Claisen-Schmidt reaction of acetone, with two equivalents of benzaldehyde? b) What is the product that results if only one equivalent of benzaldehyde is added
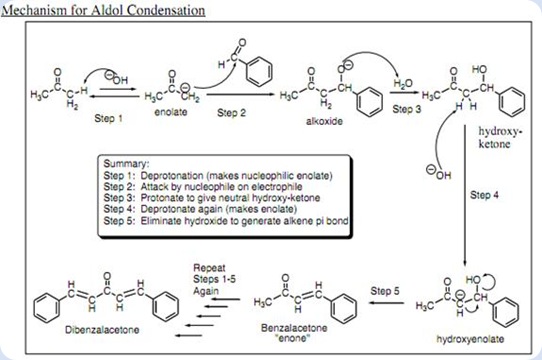
If given this balanced equation: 2C_7H_6O + C_3H_6O -> C_17H_14O +2H_2O Why is the acetone deprotonated and not the benzaldehyde? | Socratic