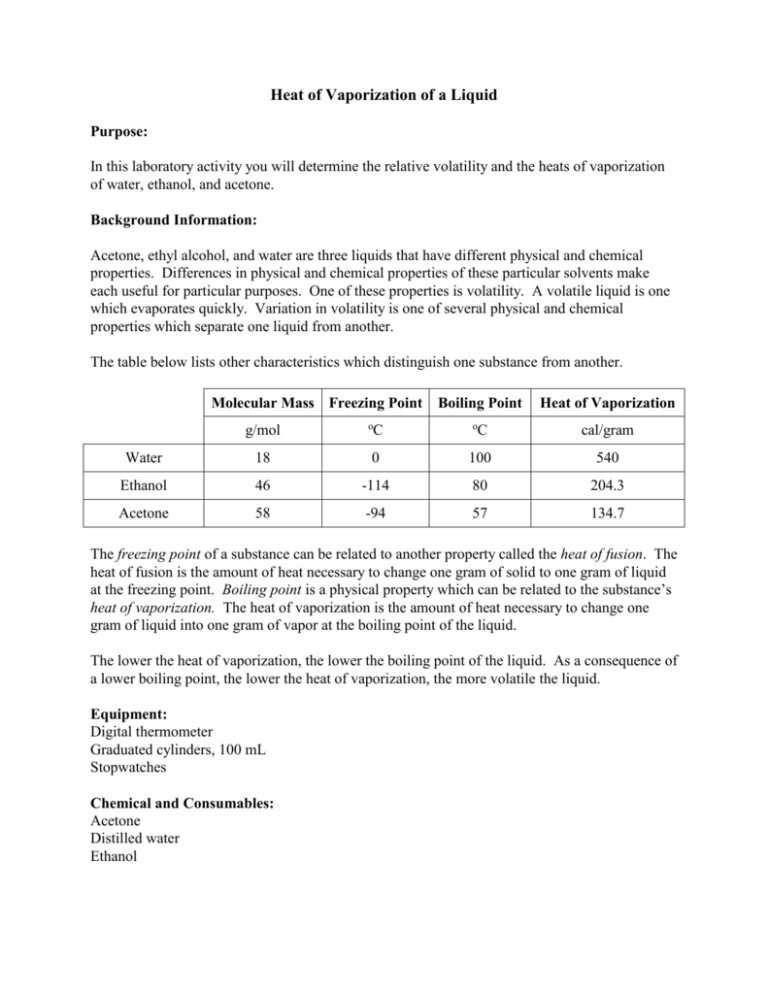
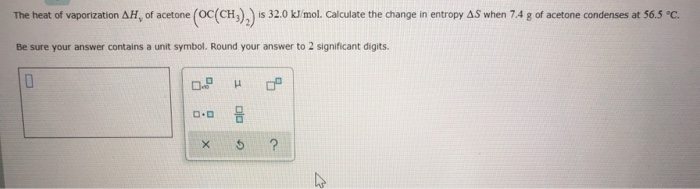
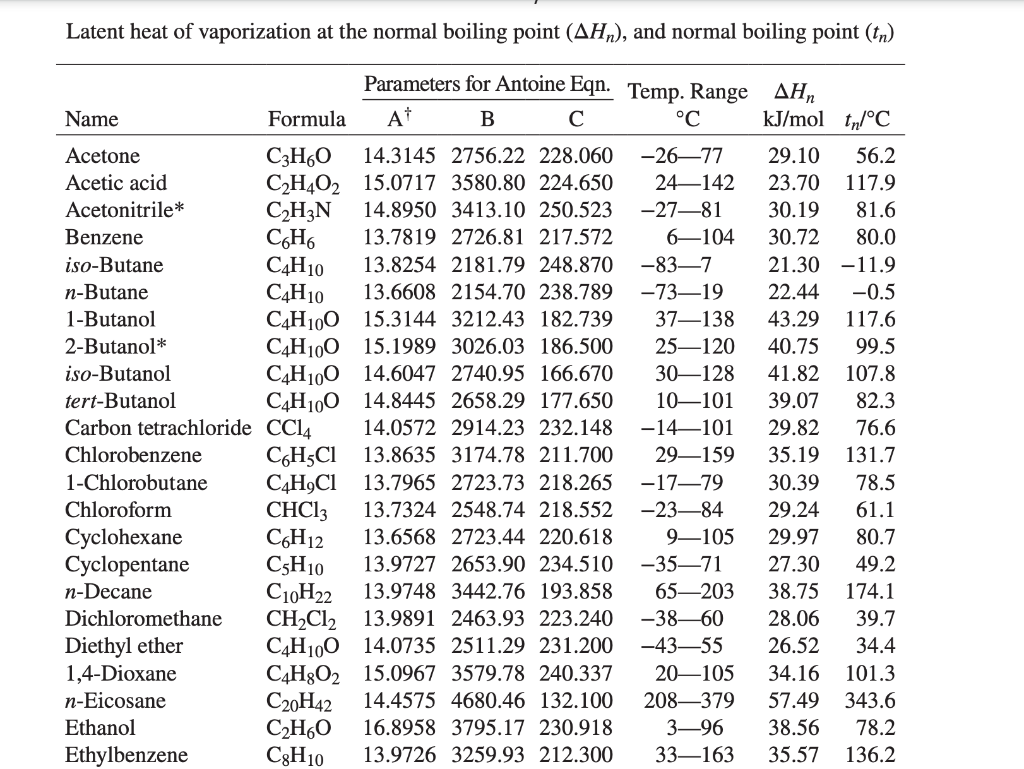
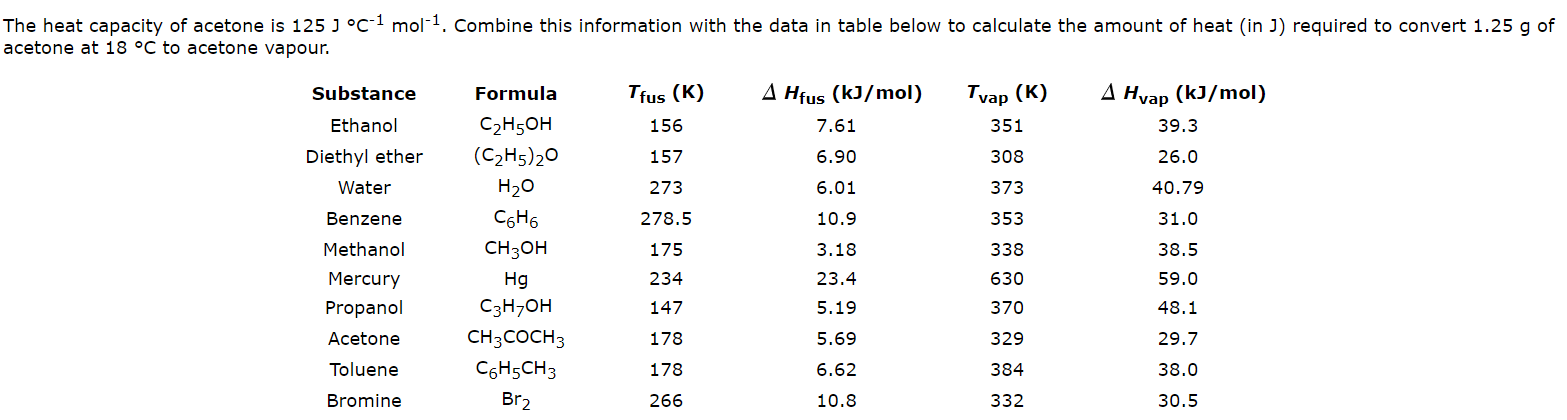
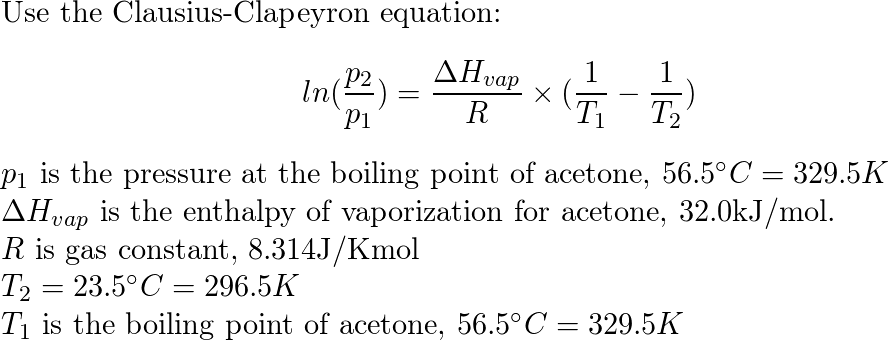SOLVED: How much heat is required to vaporize 38.5 g of acetone (C3H6O; molar weight 58.1 g) at 25 °C? The heat of vaporization for acetone at this temperature is 31.0 kJ/mol.

Ch11-ClausiusClapeyron 2 .docx - Clausius-Clapeyron equation and heat of vaporization adapted from Dr. Sushilla Knottenbelt NAMES: 1a. In order | Course Hero

The enthalpy of vaporization for acetone is 320 kJ/mol The normal boiling point for acetone is 56 - YouTube
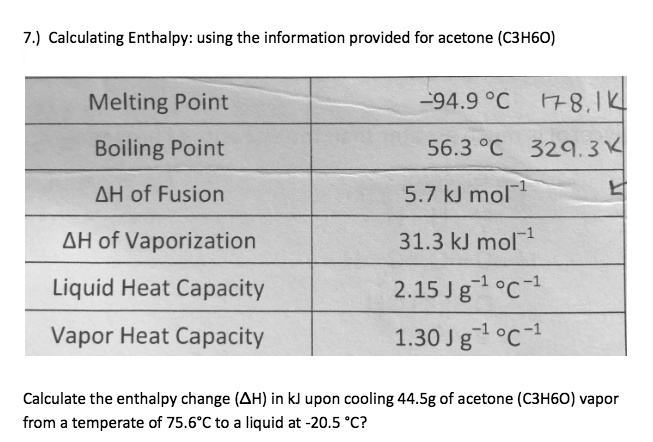
SOLVED: Calculating Enthalpy: using the information provided for acetone (C3H6O) Melting Point Boiling Point AH of Fusion 94.9 °C 56.3 °C 329.34 kJ mol-1 AH of Vaporization 31.3 kJ mol-1 2.15 J

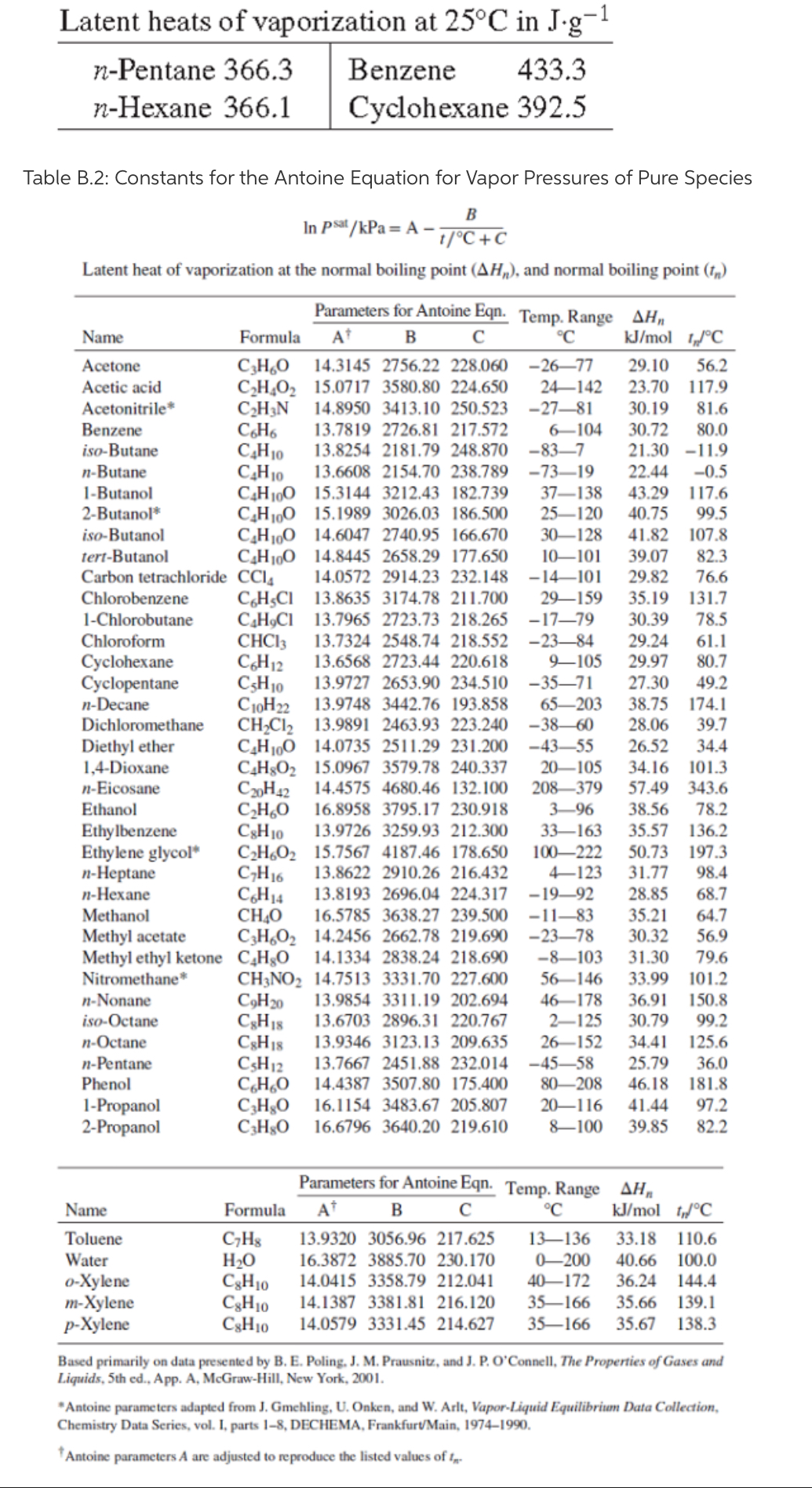
Heat of vaporization of acetone. Simulation data: • this work, AUA4... | Download Scientific Diagram

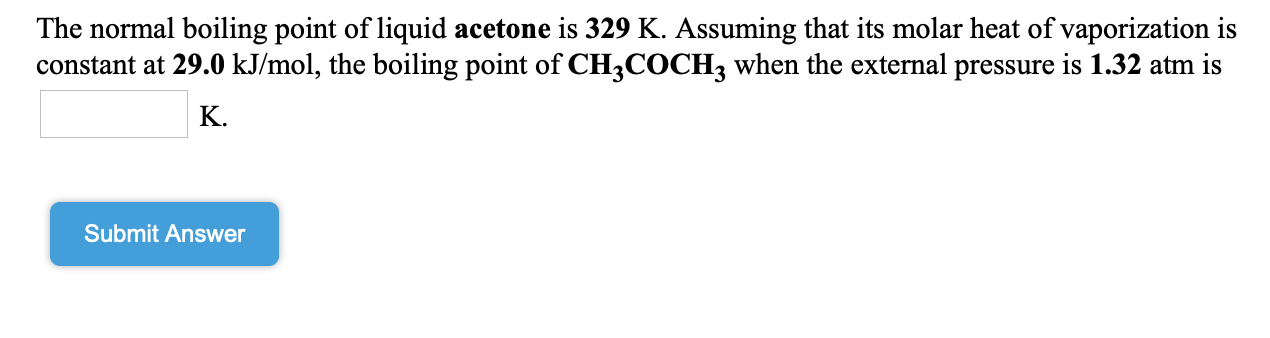
SOLVED: The normal boiling point of acetone is 56.2 °C and the molar heat of vaporization is 32.0 kJ/mol. At what temperature will acetone boil under a pressure of 40.0 mmHg? (R =

SOLVED: The heat of vaporization of acetone is 31.3 kJ/mol, and it has a boiling point of 78.6°C at 1520 mm Hg. What is the boiling point when the pressure is raised

Thermophysical Properties of Liquid Acetone, Cellulose Acetate, and... | Download Scientific Diagram
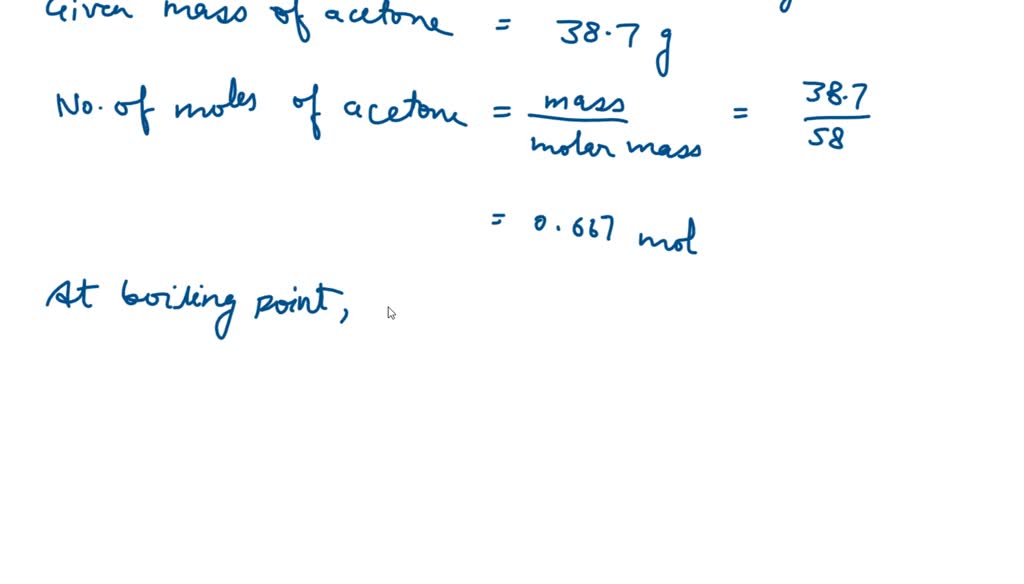
SOLVED: The heat of vaporization of acetone at its boiling point is 29.1 kJ/mol. How much energy (in kJ) do I need to add to vaporize 3.00 moles of acetone?

SOLVED: Determine the amount of heat required to vaporize 75.0 mL of acetone (C3H6O) at 25 °C. The heat of vaporization (kJ/mol) at 25 °C is 31.0. The density and the molar