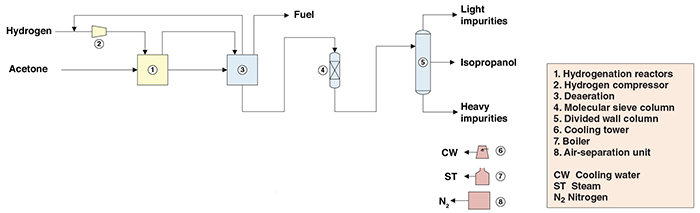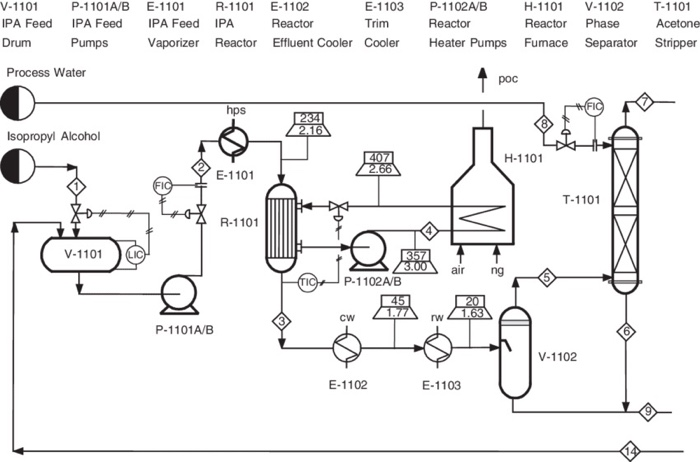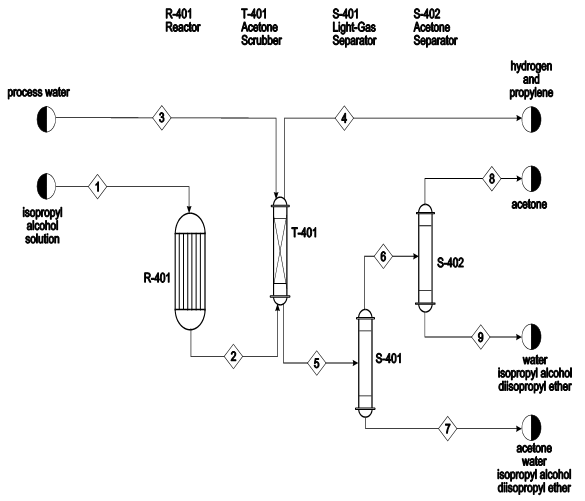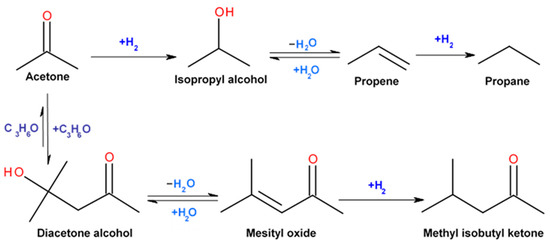
Catalysts | Free Full-Text | Highly Selective Gas-Phase Catalytic Hydrogenation of Acetone to Isopropyl Alcohol

Catalysts | Free Full-Text | Highly Selective Gas-Phase Catalytic Hydrogenation of Acetone to Isopropyl Alcohol

Acetone can be easily converted to isopropyl alcohol by addition of hydrogen to the carbon-oxygen double bond. Calculate the enthalpy of reaction using the bond energies. [{Image src='reaction22310807 | Homework.Study.com
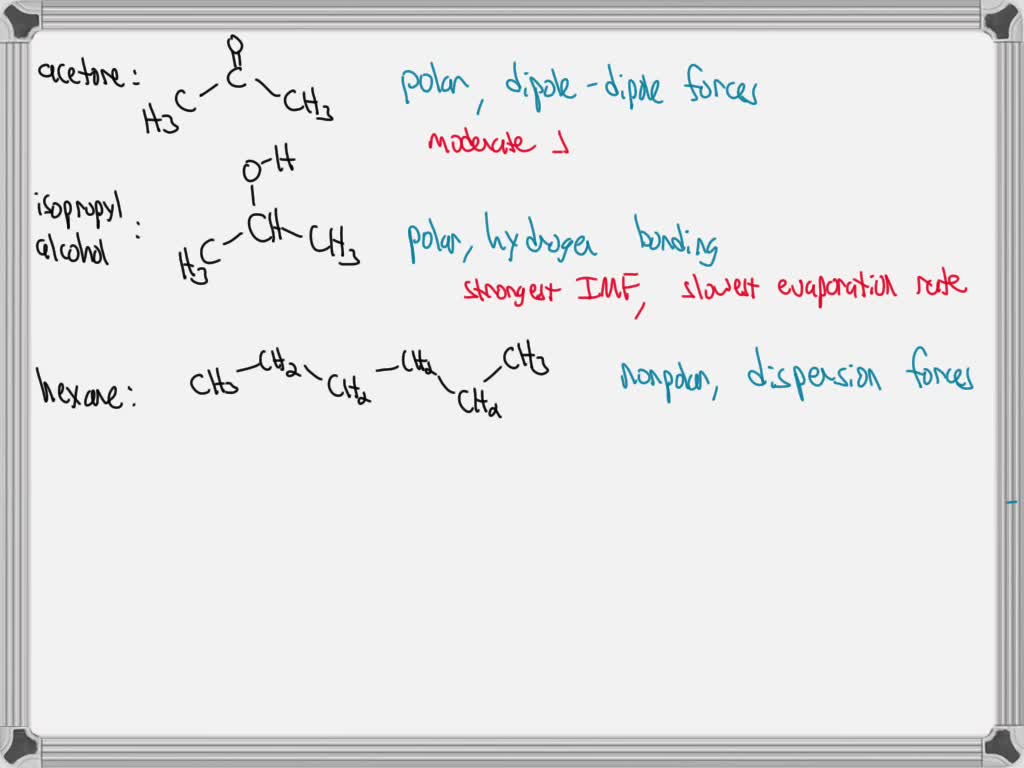
SOLVED: Using the structures of acetone, isopropyl alcohol, and hexane, explain your results for evaporation rates. For all three molecules, include the types of intermolecular forces and molecular polarity in your answer

a) Evaporation of acetone and isopropyl alcohol (IPA) and addition of... | Download Scientific Diagram